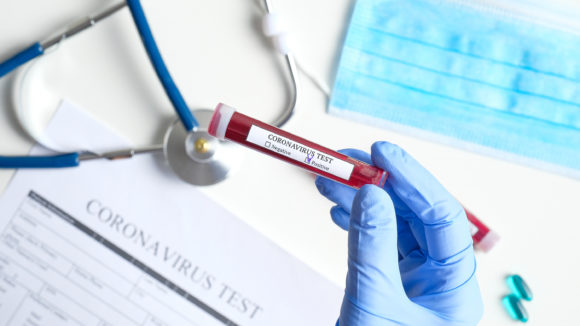
The Federal Trade Commission and U.S. Food and Drug Administration (FDA) have warned seven companies for allegedly selling unapproved products that may violate federal law by making deceptive or scientifically unsupported claims about their ability to treat coronavirus (COVID-19).
The warning is the first issued by the agencies alleging unapproved or unsupported claims that products can treat or prevent coronavirus.
The agencies said they sent letters to the following companies: Vital Silver, Quinessence Aromatherapy, N-ergetics, GuruNanda, Vivify Holistic Clinic, Herbal Amy, and The Jim Bakker Show (on the PTL Network).
According to the federal agencies, the companies have advertised products—including teas, essential oils, and colloidal silver—as able to treat or prevent coronavirus. According to the FDA, however, there are no approved vaccines, drugs, or investigational products currently available to treat or prevent the virus.
“The FDA considers the sale and promotion of fraudulent COVID-19 products to be a threat to the public health. We have an aggressive surveillance program that routinely monitors online sources for health fraud products, especially during a significant public health issue such as this one. The FDA’s laws are designed to protect the public health by ensuring, among other things, that drugs are safe and effective for their intended uses,” said FDA Commissioner Stephen M. Hahn, M.D.
The FDA said it is particularly concerned that products that claim to cure, treat or prevent serious diseases like COVID-19 may cause consumers to delay or stop appropriate medical treatment, leading to serious and life-threatening harm.
The Jim Bakker Show currently has a posting entitled,”Covid-19 Coronavirus, building immunity, staying healthy and the benefits of Silver Solution.” The FDA has previously warned that colloidal silver is not safe or effective for treating any disease or condition.
N-ergetics deleted language “per the FDA” from its website and links to the FDA letter it received with the warnings that lists multiple examples of where the FDA says false claims were made.
The warning letter to Vivify Holistic Clinic in Ontario criticizes its publishing of various claims including that “Stephen Buhner . . . has done extensive research on coronaviruses . . . He has treated them very successfully using his protocols.”
Herbal Amy has also promoted Stephen Buhner’s herbs and tea protocol that is criticized by the FDA. “Stephen Buhner has used this with other corona virus infections, including SARS, it works well,” Herbal Amy’s website says.
In the letters, the FTC alleges that one or more of the efficacy claims are unsubstantiated and therefore may violate the FTC Act. The letters advise the recipients to immediately cease making all claims that their products can treat or cure coronavirus.
The companies have been told they have 48 hours to notify the FTC of the specific actions they have taken to address the government’s concerns
The letters note that if the false claims do not cease, the FTC may seek a federal court injunction and an order requiring refunds to consumers.
Source: FTC, FDA
Was this article valuable?
Here are more articles you may enjoy.


 USI Insurance Services Claims Ex-Broker Poached Clients for Own New Agency
USI Insurance Services Claims Ex-Broker Poached Clients for Own New Agency  Helicopter Crash in Georgia Kills Groom, Pilot, Hours After Huge Wedding Celebration
Helicopter Crash in Georgia Kills Groom, Pilot, Hours After Huge Wedding Celebration  Illinois Night Club to Pay $200K in Sex and Race Discrimination Case
Illinois Night Club to Pay $200K in Sex and Race Discrimination Case  Roof Costs Soar Even as Claims Decline: Verisk
Roof Costs Soar Even as Claims Decline: Verisk 

