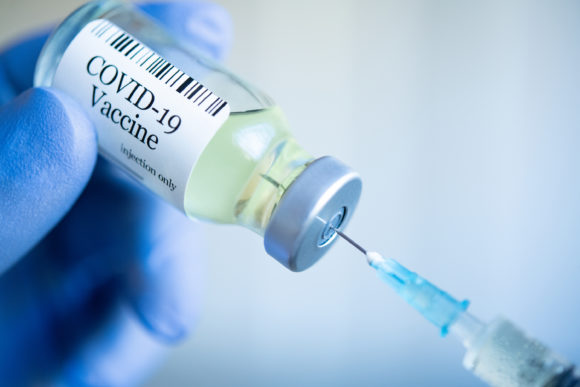
The U.S. Food and Drug Administration said on Friday it was working rapidly to issue an emergency use authorization for Pfizer Inc’s COVID-19 vaccine, with the first Americans set to be immunized as early as Monday or Tuesday.
U.S. Department of Health and Human Services Secretary Alex Azar said regulatory authorization should come within days.
A panel of outside advisers to the FDA on Thursday voted overwhelmingly to endorse emergency use of the vaccine, paving the way for the agency to authorize the shot for a country that has lost more than 285,000 lives to COVID-19.
Pfizer has asked that the two-dose vaccine, developed with German partner BioNTech, be approved for use in people aged 16 to 85.
The agency has also notified the U.S. Centers for Disease Control and Prevention and Operation Warp Speed so that they can execute their plans for timely vaccine distribution, it said in a statement.
The vaccine, which was shown to be 95% effective in preventing the disease in a late-stage trial, is already approved in Britain, and people there began receiving the shots on Tuesday.
Bahrain and Canada have also authorized the vaccine, and Canada expects to start inoculations next week.
For months, U.S. President Donald Trump had pressured the FDA in tweets and press conferences to move more quickly, accusing staff and Pfizer of dragging their feet.
Shortly after the FDA’s statement, the outgoing president criticized the agency for what he said was its slow handling of the vaccines and criticized FDA Commissioner Steven Hahn.
(Reporting by Ankur Banerjee in Bengaluru and Susan Heavey in Washington; Editing by Saumyadeb Chakrabarty)
Was this article valuable?
Here are more articles you may enjoy.


 Amazon’s Ring Sued Over Facial Recognition Feature
Amazon’s Ring Sued Over Facial Recognition Feature  USI Insurance Services Claims Ex-Broker Poached Clients for Own New Agency
USI Insurance Services Claims Ex-Broker Poached Clients for Own New Agency  Georgia Brokers and Agents Alarmed After Court Ruling Expands Liability for Them
Georgia Brokers and Agents Alarmed After Court Ruling Expands Liability for Them  Acrisure Goes After Former Owners of Businesses it Acquired for Leaving to Compete
Acrisure Goes After Former Owners of Businesses it Acquired for Leaving to Compete 

