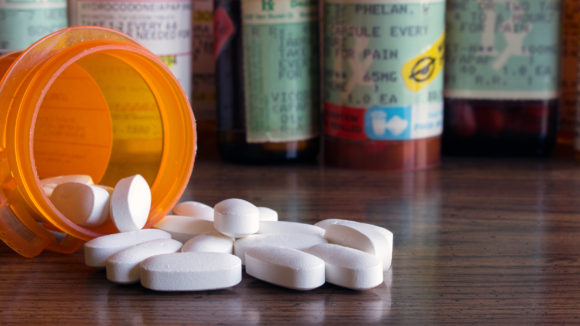
McKesson Corp.’s board reached a $175 million settlement with investors who claimed directors failed to maintain adequate internal systems for spotting suspicious opioid shipments, as the U.S.’s largest drug distributor continues to grapple with claims it helped fuel a public-health crisis tied to the painkillers.
As part of the accord, McKesson executives also agreed to beef up the company’s corporate-governance protections by separating the role of the chief executive officer and board chairman and toughening bonus claw-back policies for officials who don’t perform properly, according to court filings in California and Delaware. The money from the so-called derivative settlement — coming from directors’ insurance policies — goes into the company’s coffers.
McKesson investors overcame “steep odds to obtain a remarkable settlement,” the plaintiffs’ lawyers said last month in a motion seeking preliminary approval for the deal from U.S. District Judge Claudia Wilken in Oakland, California. A court filing in a similar case in state court in Delaware Thursday also referenced the deal.
David Matthews, a McKesson spokesman, said Thursday that while the company moved to resolve the investors’ suits, the deal “does not include any admission of liability.”
The accord comes as McKesson continues to wrestle with thousands of suits filed by states, cities and counties accusing it of contributing to the country’s opioid epidemic by failing to halt questionable shipments of the painkillers to pharmacies.
McKesson is part of a proposed settlement of the governmental suits that has been valued at about $50 billion. The deal hasn’t yet garnered enough support from state attorneys general and lawyers for municipalities though. Opponents question whether the $18 billion offer by McKesson, Cardinal Health Inc. and AmerisourceBergen Corp. is enough.
Other opioid makers, such as Teva Pharmaceutical Industries Ltd. have offered to contribute $23 billion in treatment medications and $250 million to the deal while Johnson & Johnson wants to kick in $4 billion.
Over the past decade, McKesson has twice settled with regulators who claimed it didn’t have proper systems in place to catch illegitimate orders of painkillers. McKesson has faced questioning from congressional committees about allegations it flooded small West Virginia towns with opioids.
Two years ago, a committee of McKesson directors cleared board members and senior executives of wrongdoing, saying the company “placed great emphasis on compliance” and “encouraged ethical conduct.”
‘Red Flags’
Still, Wilken rebuffed McKesson’s request to throw out the investors’ claims, saying they raise legitimate questions about whether directors ignored “multiple red flags” about opioid shipments even after agreeing to step up compliance oversight as a result of a deal with the government.
The settlement, produced in part by a series of mediation sessions, calls for McKesson to add two new independent directors to its board, to beef up compliance training for directors and improvements in internal systems designed to red-flag suspicious orders, according to court filings.
Plaintiffs’ lawyers plan to ask Wilken to approve $52 million in legal fees, which amounts to 30% of the insurance proceeds satisfying the cash portion of the deal, according to the filing.
The case is In Re McKesson Corporation Derivative Litigation, 17-cv-01850, U.S. District Court, Northern District of California (San Francisco).
Topics California
Was this article valuable?
Here are more articles you may enjoy.


 NY Lawmakers Agree to Governor’s Auto Insurance Reforms in New Budget
NY Lawmakers Agree to Governor’s Auto Insurance Reforms in New Budget  Acrisure Goes After Former Owners of Businesses it Acquired for Leaving to Compete
Acrisure Goes After Former Owners of Businesses it Acquired for Leaving to Compete  Amazon’s Ring Sued Over Facial Recognition Feature
Amazon’s Ring Sued Over Facial Recognition Feature  Travelers: Vendor Issues Over Half of Wedding Insurance Claims in 2025
Travelers: Vendor Issues Over Half of Wedding Insurance Claims in 2025 

